A common lab problem starts with a peptide that looks fine on paper. The sequence is correct, the label says high purity, the vial arrives on time, and the first assay still behaves unpredictably. One week later, a second prep gives a different result. By the time the team starts questioning the material itself, cell work, assay time, and budget have already been spent.
That's why peptide purity testing matters far beyond procurement. For research teams, purity testing is part of experimental design. It tells the lab whether the material is chemically clean, correctly identified, and present in the amount the COA suggests. Those are separate questions, and each one affects reproducibility in a different way.
For any research-use-only workflow, the safest habit is to treat the peptide COA as working analytical data, not marketing copy. Products discussed in this context are intended for research use only and not for human consumption, medical use, diagnosis, treatment, or prevention of disease.
Table of Contents
- The High Cost of Impurity in Peptide Research
- Core Analytical Techniques for Peptide Purity
- Comparing Purity Testing Methods
- How to Interpret a Certificate of Analysis (COA)
- Identifying Common Peptide Impurities
- Choosing the Right Purity Tests for Your Research
- Best Practices for Sample Handling and Documentation
The High Cost of Impurity in Peptide Research
Most failed peptide experiments don't fail dramatically, but subtly. A signaling assay gives a weak response, a binding study looks noisier than expected, or replicate sets drift apart for no obvious reason.
In many labs, the first suspicion falls on technique, instrument settings, or biological variability. Those are reasonable suspects, but reagent quality often enters the conversation too late. A peptide can carry low-level synthesis byproducts, residual process materials, or the wrong effective amount of target material in the vial, and each of those problems can distort results without producing an obvious warning sign.
A small impurity peak can matter if the experiment is sensitive to charge, hydrophobicity, aggregation behavior, or buffer conditions. The damage isn't only analytical. It affects project timelines, interpretation of mechanism, and confidence in repeat data.
Practical rule: If an experiment depends on a defined peptide concentration and sequence-specific activity, the material should be treated as an analytical input, not a commodity.
New researchers often assume that a single purity number settles the question. It doesn't. A high reported purity may still leave unanswered questions about identity, actual peptide mass, or residual non-peptidic content. That gap is where many avoidable reproducibility problems start.
The strongest labs build a habit of checking incoming documentation before the first reconstitution. They ask whether the COA includes a chromatogram, mass confirmation, and enough detail to connect the labeled vial to a specific batch result. That basic review often takes less time than troubleshooting one failed assay plate.
This is especially important in research environments where one peptide batch may feed multiple users and multiple assay formats. If the incoming quality record is weak, the uncertainty spreads through the whole project. For research-use-only materials, that uncertainty should be managed at the bench, not ignored at receiving.
Core Analytical Techniques for Peptide Purity

HPLC as the workhorse
High-Performance Liquid Chromatography, or HPLC, is the main quantitative tool in peptide purity testing. It separates components in a mixture so the lab can estimate how much of the detectable material belongs to the target peptide and how much belongs to impurity peaks.
Historically, HPLC became the gold standard for peptide purity testing in the late 1970s. By 1980, reversed-phase HPLC at 214 to 220 nm had become routine, with purity calculated as (main peak area / total peak areas) × 100%. The same development helped address synthesis quality limits. Moving from 95% to 99% pure starting materials for a 10-amino-acid peptide could raise desired product yield from about 60% to about 90%, as described in the peptide purity overview from JPT.
For a new researcher, the easiest analogy is this. HPLC is like lining up all UV-detectable components by how they interact with the column and mobile phase. The target peptide usually appears as the main peak. Smaller peaks suggest other detectable species.
That doesn't mean HPLC tells the whole story. It measures relative peak area among analytes it can separate and detect under that method. It doesn't automatically tell the lab whether the main peak is the correct sequence, nor does it reveal the true dry peptide mass in the vial.
Mass spectrometry for identity
Mass spectrometry, often paired with liquid chromatography as LC-MS, answers a different question. It asks whether the material has the expected molecular mass.
That distinction matters. A chromatogram may show one dominant peak, but mass confirmation checks whether that dominant component matches the intended peptide. In routine QC, this is one of the fastest ways to avoid working with a mislabeled or altered material.
A practical reading strategy is simple:
- Check expected mass: Compare the observed mass signal to the theoretical peptide mass.
- Check consistency with HPLC: The dominant chromatographic peak should align with the expected identity, not a secondary species.
- Look for obvious extras: Additional masses can suggest adducts, side products, or incomplete synthesis products.
A clean chromatogram without mass confirmation is reassuring, but it isn't full identity control.
LC-MS is especially useful when the lab needs both separation and identity in a single workflow. For many research settings, that combination gives enough confidence to release material into non-clinical experiments.
AAA for true peptide amount
Amino Acid Analysis, or AAA, addresses a question that new researchers often miss. How much actual peptide is present by mass?
The language surrounding purity often becomes confusing. A peptide can be highly pure by HPLC and still contain substantial non-peptidic weight from water, salts, or counterions. AAA is used to determine actual peptide content after hydrolysis and quantitative amino acid measurement.
A key practical example appears in the peptide quantitation explanation from MilliporeSigma. If a peptide is 95% pure by HPLC but has 80% content, then only 76% of the gross mass is actual target peptide. The same source notes that residual TFA is commonly present at 5 to 15% w/w, and hydration can add further non-peptide mass.
That's why AAA matters in concentration-sensitive work. It helps the lab convert vial weight into chemically meaningful peptide amount rather than assuming the label weight equals target peptide weight.
Where MALDI-TOF MS and CE fit
Two other tools appear regularly in peptide workflows.
MALDI-TOF MS is often used for rapid mass checks. It's useful when the main need is quick confirmation of a dominant molecular species. In research screening, it can support identity review efficiently, though it doesn't replace a full impurity profile.
Capillary Electrophoresis, or CE, separates species by charge-to-size behavior in an electric field. This makes it valuable for charged peptides, polar impurities, and some species that may not separate cleanly by reversed-phase HPLC. When a peptide is highly basic, hydrophilic, or prone to co-elution, CE can provide a useful second view.
A good mental model is this:
- HPLC is strongest for relative impurity profiling
- MS is strongest for identity and molecular mass
- AAA is strongest for actual peptide amount
- CE adds value for charge-based separations
- MALDI-TOF helps with rapid mass confirmation
No single method answers every quality question. Good peptide purity testing starts by matching the method to the decision the lab needs to make.
Comparing Purity Testing Methods
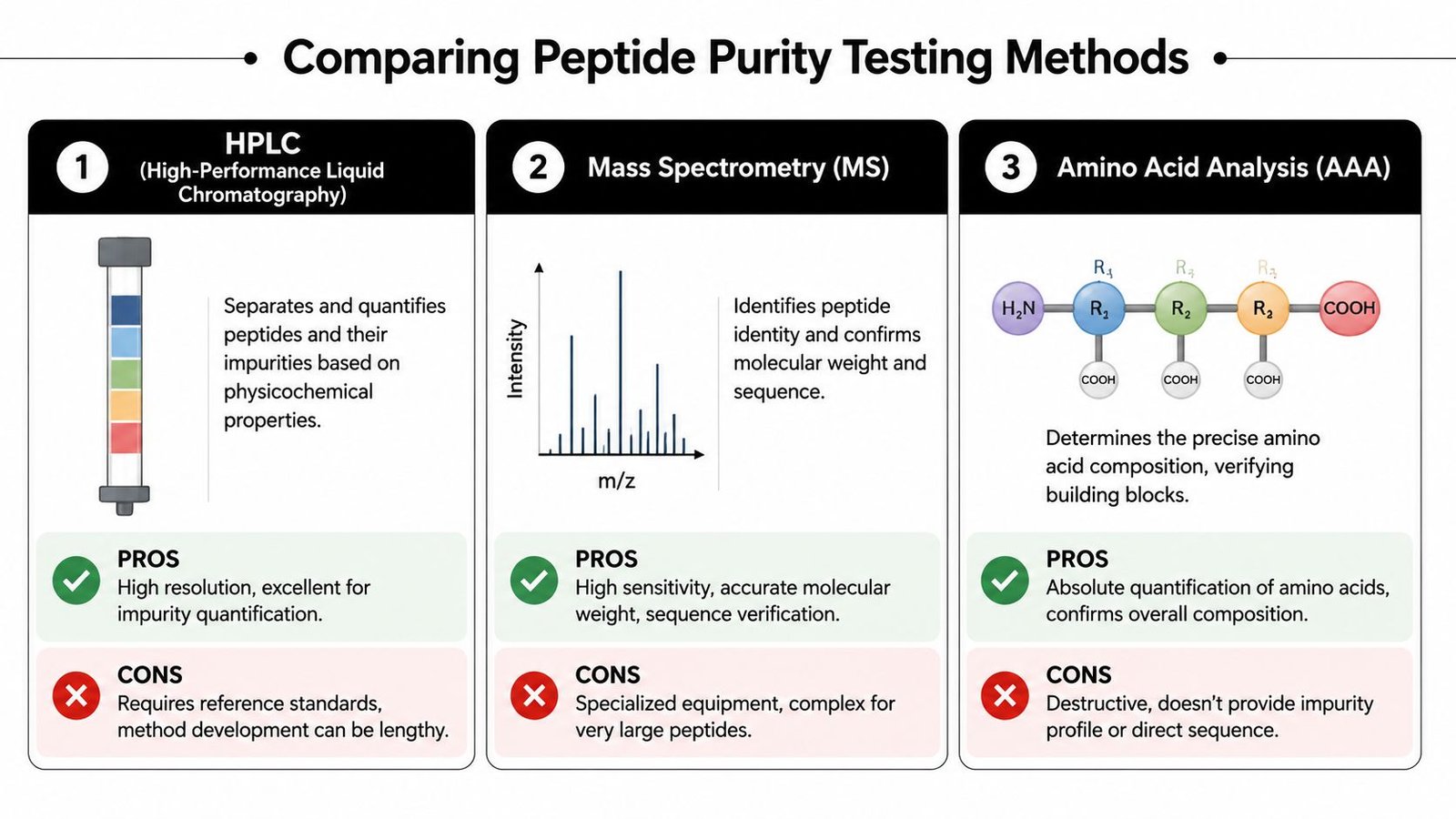
Different tools answer different questions
Researchers often ask which test is “best.” That's usually the wrong question. The better question is which test answers the specific uncertainty that could compromise the experiment.
If the concern is impurity burden, HPLC usually leads. If the concern is whether the sequence target is present, MS becomes essential. If the concern is how much peptide is really available for stock preparation, AAA becomes the deciding measurement.
The methods also differ in what they fail to show. HPLC can miss identity errors if a wrong species co-behaves under the method. MS doesn't by itself provide a full impurity distribution in the way a chromatogram does. AAA provides content, but it doesn't directly produce an impurity map or a sequence trace.
Comparison of Peptide Purity Testing Methods
| Method | Primary Purpose | What It Measures | Key Advantage |
|---|---|---|---|
| HPLC | Purity assessment | Relative distribution of target peak versus detectable impurities | Strong for impurity quantification and routine batch comparison |
| LC-MS or MS | Identity confirmation | Molecular mass of the detected species, often alongside chromatographic separation | Strong for confirming that the main species is the expected peptide |
| AAA | Content determination | Absolute peptide-related amino acid content after hydrolysis | Strong for calculating true peptide amount in the vial |
| MALDI-TOF MS | Rapid identity screening | Dominant molecular mass pattern | Fast check for expected mass profile |
| CE | Orthogonal separation | Charge-based separation of peptide-related species | Helpful for polar, charged, or difficult-to-resolve impurities |
A lab manager choosing among these methods should think in workflow terms.
For incoming routine research material, HPLC plus mass confirmation often gives a practical baseline. For troubleshooting unexplained assay behavior, the next question is often whether apparent concentration reflects real peptide amount, which points toward AAA. For unusual sequences, highly charged peptides, or persistent impurity questions, CE can help separate species that standard reversed-phase methods may compress into one region.
The decision should be tied to experimental risk, not habit. A simple binding screen doesn't need the same analytical package as a tightly controlled mechanistic study where lot-to-lot comparability matters.
How to Interpret a Certificate of Analysis (COA)

A useful COA should let a researcher answer three questions quickly. Is the peptide likely to be the right compound, how clean is it relative to detectable impurities, and how much actual peptide is present for stock preparation.
For teams handling research-use-only materials, policy awareness matters as much as analytical review. AVM Labs USA's research-use-only policy is a good example of the boundary that should stay clear in documentation and use. These materials are for laboratory research and development, not human consumption, medical use, diagnosis, treatment, or disease prevention.
Read the chromatogram first
The chromatogram is usually the fastest place to start. It shows peaks over time, with each peak representing a separated detectable component under the chosen method.
The main things to look for are straightforward:
- Main peak dominance: The target peptide should appear as the dominant peak if the reported purity is high.
- Minor peak pattern: Small surrounding peaks suggest impurity species. Their size and distribution matter more than their mere existence.
- Retention time consistency: A batch-specific retention time helps confirm that the result belongs to the lot being reviewed.
- Method context: Wavelength, column type, and solvent conditions affect what the chromatogram can and can't reveal.
A common mistake is to look only at the headline purity percentage and skip the trace itself. Two peptides can both be reported at a high purity, yet one chromatogram may show a clean baseline with a few minor peaks while another shows broad shoulders, merged regions, or asymmetric peak shape. Those patterns can matter during reconstitution and downstream assay behavior.
HPLC purity and net peptide content are not the same
This is the point that causes the most confusion in practice.
HPLC purity describes how much of the detected analyte signal belongs to the main peptide peak relative to other detected peaks. Net Peptide Content, or NPC, describes how much of the vial mass is peptide rather than water, salts, or counterions. Those are different measurements.
According to the Biosynth discussion of peptide analytics, NPC typically reduces the effective peptide fraction to about 60 to 80% of the total labeled weight, even when HPLC purity is high. That's why a peptide can appear extremely pure chromatographically while still delivering less target peptide mass than expected.
A simple analogy helps. HPLC purity is like asking whether the apples in a crate are mostly good apples rather than bruised apples. NPC is asking how much of the crate's weight is apple rather than packaging moisture and attached material. A crate can contain excellent apples and still weigh less in edible fruit than assumed.
Bench reminder: Stock solutions should be based on actual peptide content when that information is available, not on the gross vial weight alone.
Use mass balance to avoid stock solution errors
Mass balance brings the COA together. It accounts for the total composition of the vial, including target peptide, peptide-related impurities, water, and counterions.
The practical concept is simple: the vial's total mass isn't automatically equal to target peptide mass. Water and counterions add weight. If the lab ignores them, the prepared stock concentration may be overstated.
Researchers should cross-check these COA elements together:
- HPLC result for relative purity
- Mass confirmation for expected identity
- NPC or AAA-related content for actual peptide amount
- Counterion or residual component information if provided
- Lot identification linking the document to the received vial
When a COA lacks content data, that absence should be treated as uncertainty, not as evidence that content is effectively complete. That's especially important for concentration-sensitive experiments where a nominal stock concentration can drift unnoticed from the chemically real concentration.
Identifying Common Peptide Impurities
A purity percentage is only a summary. The more useful question is what kinds of impurities are present and how they are likely to affect the experiment.
Sequence-related impurities
The first category includes sequence-related impurities. These usually arise during synthesis and purification. Common examples include deletion sequences, truncations, incomplete couplings, and closely related side products.
These impurities matter because they can resemble the target peptide enough to survive purification but still behave differently in assays. A shortened sequence may retain partial binding or altered solubility. A nearly correct sequence may compete with the intended material in a way that creates noisy or misleading biological readouts.
Sequence-related impurities are often best recognized by a combination of separation and identity methods. HPLC may flag them as extra peaks. MS helps determine whether those peaks correspond to peptide-like species with masses close to the target.
Process-related impurities
The second category includes process-related impurities, such as residual counterions, moisture, and other non-peptidic materials carried through synthesis and workup.
One of the most important examples is trifluoroacetic acid, or TFA. As described in the peptide quantitation article from MilliporeSigma, TFA often remains at 5 to 15% w/w and can lower effective molarity while shifting solution pH. The same source notes that in some kinase assays, high TFA levels can cause more than 20% activity loss when pH drops below 6.
That isn't a trivial impurity. It can change assay conditions directly. A researcher may think the peptide is underperforming when the actual issue is altered solution chemistry caused by residual counterion load.
Other non-peptidic components can create similar confusion. Moisture increases gross weight. Salts can change dissolution behavior. Counterions can influence charge state and apparent handling properties.
Requesting quantification of residual components is often more useful than arguing over a single headline purity figure.
For practical interpretation, it helps to think in terms of impact:
- Wrong sequence-related species can change target engagement.
- Residual acids or salts can change solution conditions.
- Water content can distort concentration calculations.
- Low-level mixed impurities can complicate reproducibility across lots.
Peptide purity testing becomes much more informative when the lab stops treating all impurities as interchangeable. Different impurity classes create different experimental risks.
Choosing the Right Purity Tests for Your Research

A risk-based testing framework
The right testing package depends on what the peptide will do in the workflow. Not every project needs the maximum possible analytical burden, but every project does need enough evidence to control the risk of bad data.
A useful way to think about acceptance is by consequence of error.
If the peptide is being used for early exploratory work, the main concern may be obvious identity problems and major impurity burden. If the peptide will anchor a mechanism study, support lot-to-lot comparisons, or feed especially sensitive assays, then content accuracy and impurity characterization deserve more weight.
A practical framework looks like this:
- For routine incoming QC: Require a readable HPLC chromatogram and mass confirmation tied to the lot.
- For concentration-sensitive experiments: Add confidence in actual content, not just chromatographic purity.
- For difficult peptide chemistries: Consider an orthogonal method when charge-related or co-eluting species are plausible.
- For troubleshooting: Match the next test to the failure mode. Don't order every method by default.
When a full suite helps and when it doesn't
Orthogonal testing has real value, but it isn't automatically the most rational choice for every research setting.
According to this discussion of peptide purity testing in U.S. research settings, RP-HPLC can detect over 95% of common impurities for many neutral synthetic peptides, and for non-clinical research where identity is confirmed by MS, more than 98% HPLC purity can be highly reliable. The same source notes that a full analytical suite can increase cost by roughly 3 to 5 times without proportional benefit in many routine R&D cases.
That's an important correction to the usual “more testing is always better” reflex. More testing is better only when the added information changes a decision. If it doesn't, the lab may be spending budget without reducing meaningful uncertainty.
A more balanced approach involves using sufficient orthogonal evidence to cover the actual risk:
- HPLC for impurity distribution
- MS for identity
- AAA or content-focused data when concentration must be defensible
- CE only when the peptide chemistry or prior results justify it
For labs evaluating available research peptides, that framework helps separate sensible QC from overbuilt QC. The goal is reliable data, not the longest possible test list.
Questions worth asking before acceptance
A few supplier-facing questions can prevent many downstream problems:
- Does the COA include the raw chromatogram or only a summary number?
- Is mass confirmation lot-specific?
- Is net peptide content reported, or only HPLC purity?
- Are residual counterions or water discussed when relevant?
- Can the lab connect the received label directly to the batch record?
A peptide should be accepted when the documentation matches the experimental risk. If the paperwork leaves important uncertainty unresolved, the lowest-cost decision may be to pause before the first experiment starts.
Best Practices for Sample Handling and Documentation
Analytical quality can be lost after delivery if handling is sloppy. A clean batch on paper doesn't stay clean automatically at the bench.
Handling steps that protect peptide quality
Peptides should be stored and reconstituted with a plan. The exact conditions depend on the sequence and application, but the principles stay consistent.
- Control moisture exposure: Lyophilized material can pick up ambient water quickly, which changes effective mass and handling behavior.
- Limit freeze-thaw stress: Repeated cycling can promote degradation or variability in working stocks.
- Aliquot deliberately: Small, single-use aliquots reduce repeated handling of the parent material.
- Match solvent choice to peptide behavior: Solubility, charge, and downstream assay compatibility should drive the reconstitution approach.
- Mix gently but adequately: Incomplete dissolution creates concentration errors that look like biological variability.
The most common calculation error happens before the vial is even dissolved. As explained in GenScript's discussion of peptide mass balance, water is often present at 5 to 15%, and researchers can misstate stock concentration if they calculate from gross weight rather than net peptide content.
For labs that use sterile diluents in research workflows, materials such as bacteriostatic water for laboratory handling should still be selected with the experiment's compatibility and documentation needs in mind. The key point remains the same. Concentration calculations should reflect the COA's content information whenever available.
Documentation habits that save experiments
Good records are part of peptide purity testing because they preserve traceability after the vial leaves receiving.
A practical documentation checklist includes:
- Lot linkage: Record the batch or lot number in every experimental notebook entry.
- COA retention: Save the analytical PDF where the full team can retrieve it.
- Aliquot labels: Include peptide ID, concentration, solvent, date, and preparer.
- Deviation notes: If the material dissolves poorly or looks unusual, log it immediately.
The lab that can tie every result back to a specific vial and COA usually solves reproducibility problems faster.
Research-use-only materials should remain within documented laboratory workflows and are not intended for human consumption, medical use, diagnosis, treatment, or prevention of disease.
AVM Labs USA supports research teams that need documented, research-use-only peptide materials with batch-specific quality records and a professional laboratory supply experience. Researchers who want dependable peptide documentation, transparent handling standards, and a catalog built for laboratory workflows can review AVM Labs USA.
Made with Outrank app
